Biological Drugs
Claim Charts
Data Fields
IPDataLab understands the significance of data quality to our customers in the pharmaceutical industry. Thus, our PurpleBook patent database is manually curated by domain experts and measured via 4 key criteria: reliability, accuracy, depth, granularity.
RELIABLE
Each patent in our database was either actively disclosed by the owner or the subject of a term extension application, plus we also include related patents (continuation, divisional, and continuation-in-part applications) thereof, i.e., all directly linked to a drug. This supports focused research about a drug by avoiding search noise. Our data facilitates efficient Freedom to Operate analysis by providing convenient access to key patent information about each drug.
ACCURATE
Patent expiry date is of ultimate importance for pharmaceutical companies because it determines the generic entry date of a drug. In our data system, a sophisticated algorithm produces the most accurate patent expiry date possible, taking into consideration patent term extension (PTE), patent term adjustment (PTA), terminal disclaimers, etc.
IN-DEPTH
New metadata fields not available in the standard PurpleBook are provided. This metadata conveniently enables search across all fields, which in turn supports deep analytical insights. For example, we extract biological target and indication from drug label documents, and for each extended patent, not only requested extension days but also granted extension days are provided.
GRANULAR
Both approved drugs and patents are linked in our system on the most granular level. New indications of an existing drug can be found via the indication-approval date mapping, which offers a quick overview of drug development trends. For extended patents, specific claims are identified, and a claim chart mapping claims of a patent to a drug explains why the claims read on the drug product, thereby enabling a quick drill down to the most critical information.
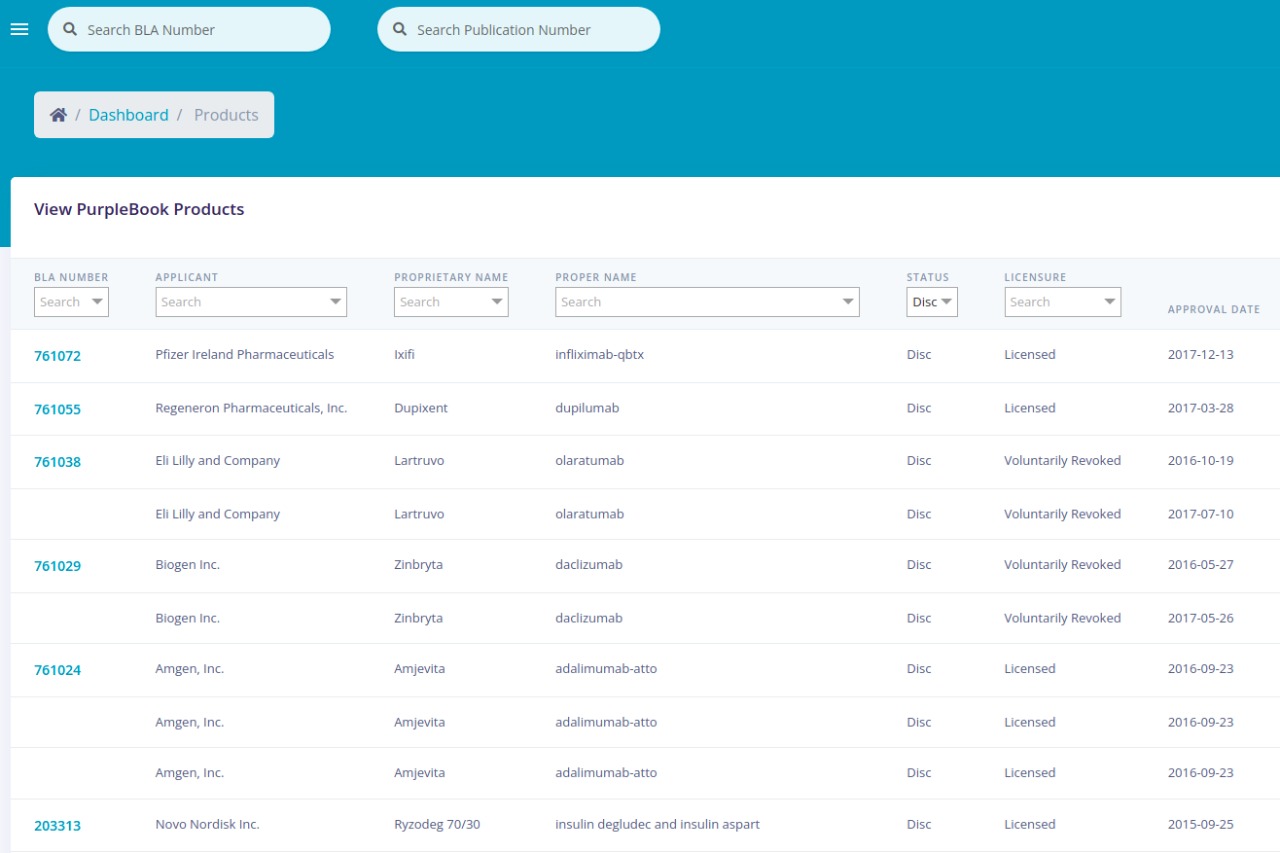
| Application Number | The number assigned by FDA staff to each Biologics License Application (BLA) for approval to market a biological product in the United States. |
| Product | The trademark or brand or proprietary name. |
| Applicant | The manufacturer or company that submits an application to FDA for approval of a drug, including a biological product, in the United States. |
| Active Ingredient | The nonproprietary name designated by FDA in the license for a biological product licensed under the PHS Act. The nonproprietary name means a name unprotected by trademark rights that is in the public domain. |
| Type | The type of a Biologics License Application (BLA), either 351(a), 351(k) biosimilar, or 351(k) interchangeable. |
| Strength | The amount of active ingredients in the dosage form of a drug |
| Dosage Form | The physical form that delivers a dose of the drug, such as a tablet, a capsule, or an injection. |
| Route of Administration | A way of administering a drug to a site in a patient (for example, intravenous or subcutaneous). |
| Product Presentation | The form of a drug that is presented by the manufacturer, e.g., Single-Dose Vial, Autoinjector, Pre-Filled Syringe, Pouch. |
| Status | Status indicates how a biological product is sold in the United States: Prescription (Rx), Over the Counter (OTC), Discontinued (Disc). |
| Licensure | Status of the product license, e.g., whether the product is currently licensed or whether the license has been revoked. |
| Approval Date | The date FDA approved the application or supplement. |
| Ref. Product Active Ingredient | The active ingredient of the single biological product, licensed (approved) by FDA under section 351(a) of the PHS Act, against which a proposed biosimilar or interchangeable product is compared. |
| Ref. Product | A reference product is the single biological product, licensed (approved) by FDA under section 351(a) of the PHS Act, against which a proposed biosimilar or interchangeable product is compared. |
| Submission Type | The type of submission, for example, the original application or a supplemental application. |
| Submission | Document number of the submission, e.g., Orig-1, Suppl-3 |
| Supplement Number | Manufacturers may submit supplements to request approval for a change to an approved BLA. A number is assigned to each supplement which is usually, but not always, sequential, starting with 001. |
| License Number | The number of the license granted to the manufacturer by the FDA. |
| Product Number | A product number is assigned to each biological product associated with a BLA (Biologics License Application). For example, if the biological product is available in multiple strengths, there are multiple product numbers. |
| Date of First Licensure | The date on which any applicable reference product exclusivity begins to run. FDA has not made a determination of first licensure for each 351(a) biological product included in the Purple Book. The absence of a date of first licensure in the Purple Book does not mean that a biological product on the list is not, or was not, eligible for the periods of exclusivity described in section 351(k)(7) of the PHS Act. |
| Center | Refers to either FDA's Center for Biologics Evaluation and Research (CBER) or Center for Drug Evaluation and Research (CDER). |
| Exclusivity Date | The date on which all applicable periods of exclusivity expire. |
| Ref. Product Exclusivity Expiration Date | The date on which the 12-year period of reference product exclusivity expires, including any applicable pediatric extension. |
| First Interchangeable Exclusivity Expiration Date | The date a first interchangeable product’s period of exclusivity ends. |
| Orphan Exclusivity Expiration Date | The latest date on which a period of seven-year period of orphan exclusivity expires, including any applicable pediatric extension. |
| Patent Number | A list of all patents identified for a drug. |
| Expiry Date | The expiry date of an idenfied patent, taken into account of all relevant factors including PTA, PTE, terminal disclaimer, etc. |
| Related Applications | The Continuation, Divisional, and CIP applications of the patents identified for a drug. |
| Target | A drug target is a molecule in the body, usually a protein, that is intrinsically associated with a particular disease process and that could be addressed by a drug to produce a desired therapeutic effect. |
| Indication | An indication of a drug refers to the use of that drug for treating a particular disease. All indications approved by FDA on a drug are listed. |
| Indication-Approval Date Mapping | Map the Approval Date to each new indication on the same drug in a table view. |
| PTA | Patent Term Adjustment (PTA), which compensate for delays caused by USPTO during prosecution. |
| Requested PTE | The number of days requested for Patent Term Extension (PTE) by patentee on the patent related to a drug under 35 U.S.C. §156. |
| Granted PTE | The number of days indicated in the Patent Term Extension Certificate on the patent related to a drug. |
| PTE Status | PTE status indicates the status of a PTE application on the patent: requested, withdrawn, denied, or approved. The PTE status is updated from time to time until the final determintion or PTE certificate is issued. |
| Claim Chart | Map the claims of a patent to a drug in a table view, explaning why the claims read on the drug. |
| Relevant Claims | The claim # of a patent that is specifically relevant to an approved drug. |
| TD Reference To | The patents referenced in a Terminal Disclaimer document, which disclaimed the term of the present patent beyond these referenced patents. |
| TD Reference By | The present patent that disclaimed the term beyond the referenced patents. |
| Bulk download Label PDF | Allow for bulk downloading drug Label PDF documents for the original approval and additional indications of a drug. |
| Bulk download PTE_related PDF | Allow for bulk downloading Patent Term Extension (PTE) related file history documents. Such documents include PTE application, the correspondence between USPTO and FDA, Notice of Final Determination, Election amongs multiple applicable patents, PTE Certificate, etc. |

This option is for customers that desire to immediately search and interact with this data set in an online research platform hosted by us, IPDataLab.
Select this option for electronic payment support leading to immediate access to the IPDataLab research platform.
Subscribe Now
This option is for customers that desire to integrate some or all of this data set into their own commercial or research system. In most cases, redistribution of the data, even commercially, is not prohibited.
Select this option to access a contact form and send us a request for pricing.
Contact for pricing.Let us show you what is possible with leading edge intellectual property data intelligence.
Get In Touch